
Image by Galarza Creador from
15 Fun Facts about Atoms
Atoms may be small, i.e. a single grain of sand contains around 22 billion atoms but they hold a wealth of fascinating secrets that continue to captivate scientists and non-scientists alike. Atoms are the building blocks of everything around us, from the air we breathe to the devices we use every day. They are incredibly small and yet they have a tremendous impact on our world. For instance, the human body is made of a collection of atoms that make up the matter of the human body. They are in different states, the blood is a liquid, the bone is solid and the air we breathe in and out is a gas.
Every element has a unique atomic number, which is determined by the number of protons in its nucleus. If you take any object and keep breaking them into smaller particles, at the end of the process, you are left with science atoms. Here are 15 fun facts about atoms that will give you a glimpse into their fascinating nature and the role they play in our lives. From the complexity of their structures to their strange behaviours, these facts will spark your curiosity and deepen your appreciation for the amazing world of atoms.
1. The Basic unit of Matter

Image by Christinelmiller from
Atoms are the basic units of matter and are made up of protons, neutrons, and electrons. Atoms are the fundamental components of everything we see and touch, and they are the fundamental building blocks of matter. Even though these tiny particles are so small that they can’t be seen with the naked eye, they still have a lot of power and complexity. Atoms combine to form molecules, compounds, and all of the substances that make up our world. Each atom has a unique combination of positively charged protons, neutral neutrons, and negatively charged electrons. These amazing building blocks make up the very fabric of our existence, from the air we breathe to the food we eat. There would be no chemistry, biology, or life as we know it if there weren’t atoms.
55 Most Amazing Scientists who are Still alive Today.
2. Atoms, Indivisible?….Mmmmh!
The ancient Greek word “atomos,” meaning “indivisible,” is the source of the word “atom.” This was due to the widespread belief among early scientists that atoms were the smallest possible units of matter and that they could not be divided further. Around 400 BCE, the Greek philosopher Democritus used this term to describe the smallest possible particle. He believed that these indivisible particles, which he referred to as atoms, were the building blocks of matter. Nevertheless, it is important to keep in mind that Democritus’ concept of atoms was distinct from the current understanding of atoms in physics and chemistry.
While modern atoms can be divided and are not indestructible, Democritus held that atoms were indivisible and indestructible. The true nature of atoms wasn’t fully understood until the late 19th and early 20th centuries when the modern atomic theory was developed. We now know that nuclear reactions can split atoms into smaller particles.
Read This: Facts about Geneva Science Museum.
3. Fun Fact: Atoms are Always in Motion!

Image by A Greg from
Atoms are always in motion but this movement is not random and is governed by the laws of physics. Atoms vibrate and move even when they are at rest, and that energy appears as heat energy. This motion increases as temperature increases, causing atoms to move faster and collide with each other. In fact, atomic motion is responsible for many observable phenomena, such as the expansion of materials when heated and the diffusion of molecules in solution. The ability to manipulate and control the motion of atoms has also led to breakthroughs such as the development of computer chips and nanotechnology. In short, the constant motion of atoms is a fundamental aspect of the universe we live in, shaping everything from how matter behaves to how our bodies function.
4. Who are the Heavy Hitters of the Atomic World?

A billet of highly enriched uranium. Image by Unknown Author from
Did you know that uranium is the heaviest atom that can be found naturally? This fun fact about atoms demonstrates how complicated the universe is and how much more there is to learn about it. Uranium is a naturally occurring element with an atomic number of 92. It is the heaviest natural atom due to its 92 protons and 146 neutrons. With a half-life of 4.5 billion years, it is also one of the radioactive elements! Uranium, with an atomic weight of 238.02, is the element that occurs naturally in the greatest quantity.
Oganesson, on the other hand, is the heaviest synthetic chemical element, having an atomic number of 118 and the symbol Og. At the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, a team of American and Russian scientists first synthesized it in 2002. Oganesson is a noble gas with a very short half-life, making it challenging to investigate its properties. In 2006, the International Union of Pure and Applied Chemistry (IUPAC) confirmed its discovery.
5. As Light as a Feather…or is it?
The lightest element is hydrogen, which has only one proton and one electron. It is the lightest and simplest element in the periodic table, with an atomic number of 1. It is a colourless, odourless, and tasteless gas that is highly flammable and reactive. The expression “hydrogen is lighter than a feather” is often used as an analogy to convey how light hydrogen is, but it is not entirely correct. However, if you compare the weight of one hydrogen atom to the weight of a feather, feathers are much heavier. A hydrogen atom has a mass of about 1.67 × 10^-27 kg, which is incredibly small compared to the weight of a tiny feather. The saying “hydrogen is lighter than a feather” isn’t entirely true, but it’s a valid way to convey just how light hydrogen really is.
Next Read: 15 Brightest Scientists of all Time.
6. Classification of Atoms

Image by from
The Periodic Table is a fundamental tool for understanding the structure of atoms and the elements that make up our universe. It is a tabular arrangement of all the chemical elements in order of their atomic number, electron configurations, and chemical properties. The table is arranged in rows and columns, with the elements placed in order of increasing atomic number from left to right across each row. It was first developed by the Russian chemist Dmitri Mendeleev in 1869 and has been refined over the years as new elements have been discovered and the properties of existing elements better understood. It is an essential part of chemistry and physics, and it has been used for centuries to help scientists understand the properties of different elements.
Each row is called a period, and each column is called a group or family. Elements in the same group have similar chemical and physical properties because they have the same number of valence electrons. The modern Periodic Table consists of 118 elements, including 94 naturally occurring elements and 24 synthetic elements that have been created in laboratories. The elements are organized into four blocks: s-block, p-block, d-block, and f-block, based on the electron configuration of their outermost energy level.
7. Fun Fact: The Human Body is made up of Octillions of Atoms

Image by from
Did you know that the human body is made up of billions of atoms? Atoms are the building blocks of all matter, and they make up everything from the air we breathe to the food we eat. A strand of human hair which is 100 nanometers across consists of one million carbon atoms. The human body is primarily composed of six elements: oxygen, carbon, hydrogen, nitrogen, calcium and phosphorus. These elements combine to form various molecules, such as proteins, carbohydrates, fats, and DNA that are essential for life. It is estimated that the human body is composed of approximately 7 octillion atoms (that’s 7 followed by 27 zeros!), although the exact number may vary depending on the individual’s size and composition. The fun fact is that you renew about 98% of them every year without even realizing it! The fastest-changing molecule is water as almost 50 % of water molecules in the body are replaced every 8 days.
Related Read: 20 Wierd Facts about the Human Body.
8. Small Size but Vast Emptiness
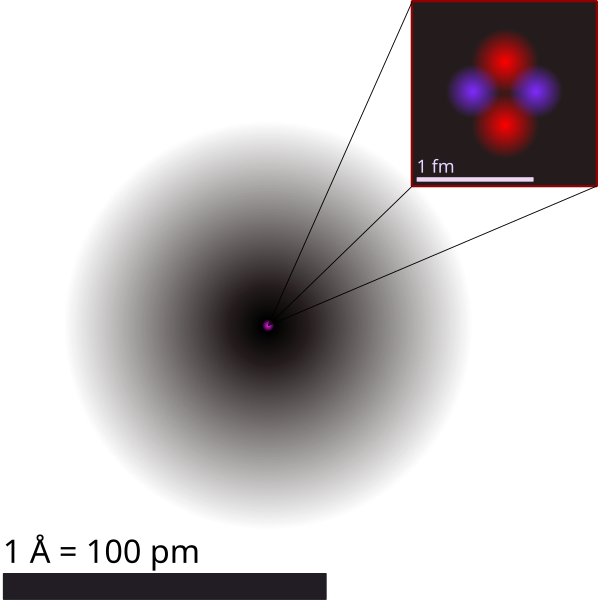
Image by Yzmo from
Picture this, if you were to enlarge a hydrogen atom to the size of a football stadium, the nucleus would be about the size of a marble at the centre, while the electrons would be like small particles orbiting the stadium’s outer edges. Atoms are mostly empty spaces. The nucleus of an atom is extremely dense and contains nearly all of the mass of each atom. Atoms consist of a nucleus at the centre, which is made up of protons and neutrons, and electrons that orbit around the nucleus.
Although the nucleus is much denser compared with the rest of the atom, it too consists mainly of empty space. The size of the nucleus is incredibly small compared to the size of the entire atom the electrons that orbit around the nucleus occupy a relatively large amount of space but have very little mass, which contributes to the overall emptiness of the atom. It takes 1,836 electrons to equal the size of a proton and orbit so far away from the nucleus that each atom is 99.9% empty space.
9. How Many Kinds of Atoms are there?
Each atom has a unique combination of three particles protons, neutrons, and electrons, which determines its properties and characteristics. There are over 118 known elements in the periodic table, each with its own type of atom. Rather elements are organized on the periodic table based on their atomic number, which is the number of protons in the nucleus of an atom. This means that there are potentially millions of different kinds of atoms in existence! The first 92 elements occur naturally, while the remaining elements are synthetic and have been created in laboratories. Each element has unique properties and can combine with other elements to form compounds and molecules, which are the building blocks of all matter. Atoms can also be changed by undergoing chemical reactions and combining them to make molecules.
Career in Science Insights: Things to Know about Medical Laboratory Scientist.
10. What Holds the Particles of Atoms Together?

Nuclear Explosion. Image by from
The components of an atom are held together by three forces. Protons and neutrons are held together by strong and weak nuclear forces. Electrical attraction holds electrons and protons. While electrical repulsion repels protons away from each other, the attractive nuclear force is much stronger than electrical repulsion. Protons and neutrons are held together by a strong nuclear force that binds them together in the nucleus of an atom. This force is so strong that it can hold protons and neutrons together even when they have opposite charges. The nuclear force is responsible for keeping atoms stable and allowing them to form molecules, which make up all matter in the universe.
The strong nuclear force is one of the four fundamental forces in nature the others being gravity, electromagnetism and the weak nuclear force. This nuclear force is about 1,038 times stronger than the gravitational force but only operates on a very small scale. The strong force that binds together protons and neutrons is 1,038 times more powerful than gravity, but it acts over a very short range, so particles need to be very close to each other to feel its effect.
11. Itsy Bitsy; it is how they Roll
Atoms are so tiny that even with most microscopes and the naked eye, they are impossible to see. Because of how small they are, their size is typically measured in nm, which is one billionth of a meter. A typical atom has a diameter between 0.1 and 0.5 nanometers, depending on the element. To put this into perspective, human hair has a diameter of about 100,000 nanometers, which means that millions of atoms could fit across the width of one human hair. Atoms are the building blocks of all matter and are necessary for comprehending the nature of the physical world around us despite their small size.
12. …….Fun Fact: Atoms can be Seen

An ESR-STM at the Center for Quantum Nanoscience in Ewha Womans University in Seoul. Image by Rickinasia from
Atoms are too small to be seen with visible light or conventional microscopes, so we can’t see them directly with our eyes. However, scientists have developed specialized instruments and techniques that allow us to observe and study atoms indirectly. One such technique, developed in the 1980s, is called scanning tunnelling microscopy (STM). STM uses a sharp needle to scan the surface of a material and measures the current flowing between the stylus and the material. By analyzing these electrical signals, scientists can create detailed images of the material’s surface, including individual atoms.
The second method, called atomic force microscopy (AFM), is similar to STM but uses a different method of detecting the presence of atoms. AFM works with a small probe that interacts with the surface of the material and measures the force between the probe and the atoms. By analyzing these forces, the scientist can create a three-dimensional image of the material’s surface, including individual atoms.
What does it take to have a career in Quantum Research? Click Here!
13. Biggest and Smallest Atom
The largest atom is the cesium atom, which has 55 electrons and 6 energy levels. It has an atomic radius of approximately 298 picometers (pm). The largest element (in terms of size) is Francium, but since it is extremely unstable, the prestige goes to cesium. It has a big valence shell and a relatively less effective nuclear charge. The smallest atom is the hydrogen atom, which consists of only one proton in its nucleus and one electron orbiting around the nucleus. It has an atomic radius of approximately 25 picometers (pm). However, it is important to note that the size of an atom can vary depending on the method used to measure it, as well as its ionization state and other factors.
14. Atom + Atom = Elements

Image by from
Different types and numbers of atoms fit together to make different elements and combine in different ways to make different types of matter, all with their own unique number of protons, neutrons, and electrons. The number of protons in the nucleus of an atom determines which element it is, and the number of electrons in the atom determines its chemical behaviour. For instance, oxygen (O) consists of eight protons, eight neutrons, and eight electrons, while the most abundant element in the universe Hydrogen (H) consists of one proton and one electron.
15. And the Atom Census Report States…….
The observable universe is vast: it spans approximately 93 billion light-years. As per the theoretical estimation, there are 1078 to 1082 atoms in our universe. It is impossible to determine the exact number of atoms in the universe because the universe is constantly expanding and our current observations only allow us to see a small portion of it. However, scientists estimate that there are approximately 10^80 atoms in the observable universe. To put this number into perspective, consider that the average human body contains about 7 octillion (7 x 10^27) atoms. The number of atoms in the universe is so large that it’s difficult to even conceptualize, but it gives us a sense of the vastness and complexity of our universe. More accurate numbers will be available as we learn more about the cosmos.
Though the atom is not visible to us it governs all the properties of matter which is why there is a need to care about atoms and increase study on them. Atoms are fascinating and complex building blocks of matter that makeup everything we see and experience in the world around us. While they are incredibly small, they are essential to our understanding of the universe.
Planning a trip to �鶹APP ? Get ready !
These are ���������Dz�’�����������-����������Բ� travel products that you may need for coming to �鶹APP.
Bookstore
- The best travel book : Rick Steves – �鶹APP 2023 –��
- Fodor’s �鶹APP 2024 –��
Travel Gear
- Venture Pal Lightweight Backpack –��
- Samsonite Winfield 2 28″ Luggage –��
- Swig Savvy’s Stainless Steel Insulated Water Bottle –��
We sometimes read this list just to find out what new travel products people are buying.









